AB
Chapter 2: Properties of Matter 41
VOCABULARY
physical property p. 41
density p. 43
physical change p. 44
chemical property p. 46
chemical change p. 46
BEFORE, you learned
• Matter has mass and volume
• Matter is made of atoms
• Matter exists in different states
NOW, you will learn
• About physical and chemical
properties
• About physical changes
• About chemical changes
KEY CONCEPT
Matter has observable
properties.
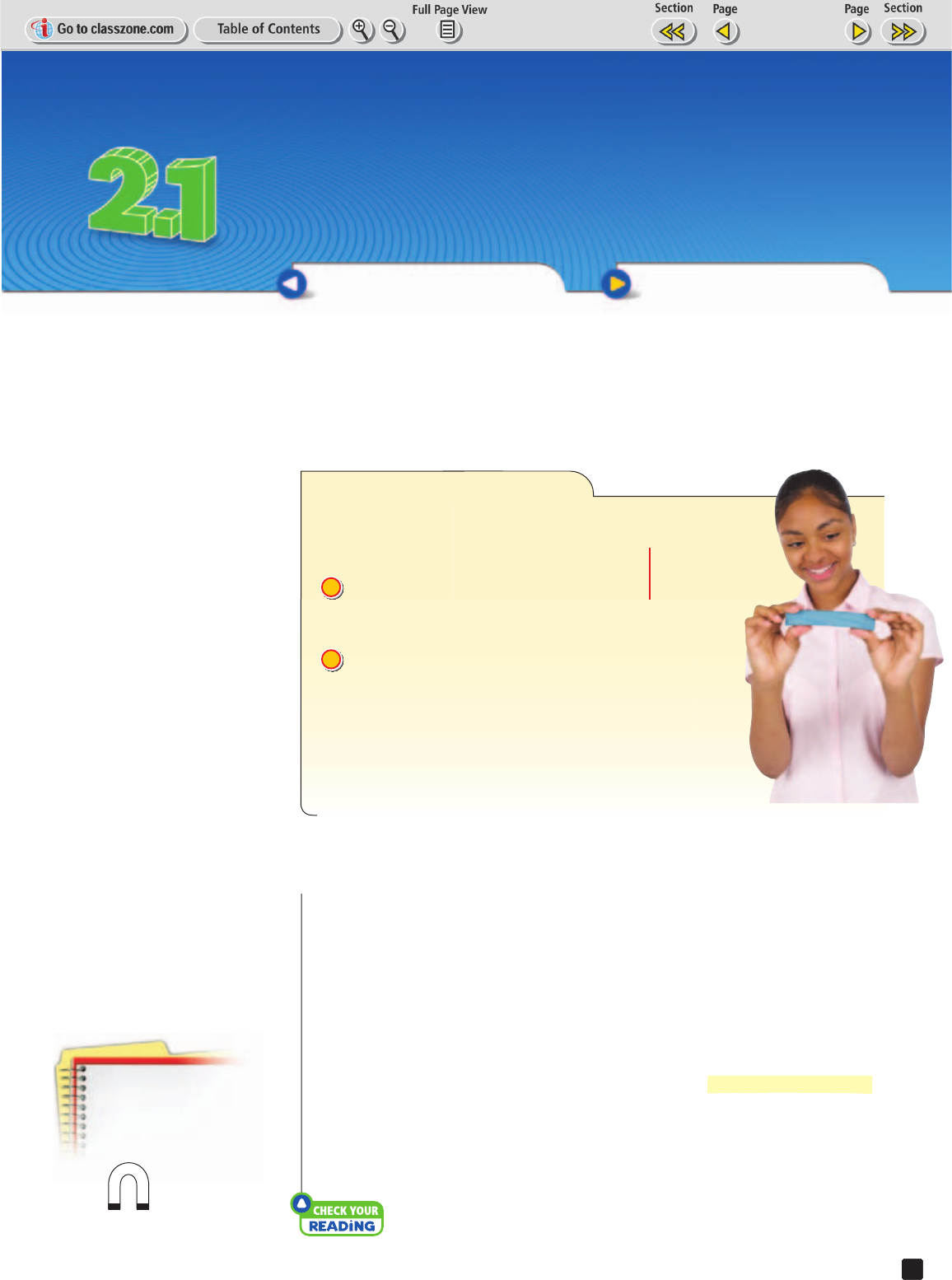
EXPLORE Physical Properties
How can a substance be changed?
PROCEDURE
Observe the clay. Note its physical
characteristics, such as color, shape,
texture, and size.
Change the shape of the clay. Note which
characteristics changed and which ones
stayed the same.
WHAT DO YOU THINK?
• How did reshaping the clay change its physical
characteristics?
• How were the mass and the volume of the clay affected?
2
1
MATERIAL
rectangular
piece of clay
Physical properties describe a substance.
What words would you use to describe a table? a chair? the sandwich
you ate for lunch? You would probably say something about the shape,
color, and size of each item. Next you might consider whether it is hard
or soft, smooth or rough to the touch. Normally, when describing
an object, you identify the characteristics of the object that you can
observe without changing the identity of the object.
The characteristics of a substance that can be observed without
changing the identity of the substance are called
In science, observation can include measuring and handling a substance.
All of your senses can be used to detect physical properties. Color, shape,
size, texture, volume, and mass are a few of the physical properties you
probably have encountered.
Check Your Reading Describe some of the physical properties of your desk.
physical properties.
VOCABULARY
Make a magnet word
diagram in your notebook
for physical property.
Page 1 of 8

AB
42 Unit: Matter and Energy
Physical Properties
How do you know which characteristics are physical properties?
Just ask yourself whether observing the property involves changing
the substance to a different substance. For example, you can stretch a
rubber band. Does stretching the rubber band change what it is made
of? No. The rubber band is still a rubber band before and after it is
stretched. It may look a little different, but it is still a rubber band.
Mass and volume are two physical properties. Measuring these
properties does not change the identity of a substance. For example,
a lump of clay might have a mass of 200 grams (g) and a volume of
100 cubic centimeters (cm
3
). If you were to break the clay in half, you
would have two 100 g pieces of clay, each with a volume of 50 cm
3
.
You can bend and shape the clay too. Even if you were to mold a
realistic model of a car out of the clay, it still would be a piece of clay.
Although you have changed some of the properties of the object, such
as its shape and volume, you have not changed the fact that the sub-
stance you are observing is clay.
Check Your Reading Which physical properties listed above are found by taking
measurements? Which are not?
reminder
Because all formulas
for volume involve the
multiplication of three
measurements, volume
has a unit that is cubed
(such as cm
3
).
Physical properties of clay—such as volume, mass, color, texture,
and shape—can be observed without changing the fact that the
substance is clay.
Physical Properties
Block of Clay Shaped Clay
COMPARE AND CONTRAST Which physical properties do the
two pieces of clay have in common? Which are different?
Page 2 of 8
AB
Chapter 2: Properties of Matter 43
Density
The relationship between the mass and the volume of a substance is
another important physical property. For any substance, the amount
of mass in a unit of volume is constant. For different substances, the
amount of mass in a unit of volume may differ. This relationship
explains why you can easily lift a shoebox full of feathers but not one
filled with pennies, even though both are the same size. A volume of
pennies contains more mass than an equal volume of feathers.
The relationship between mass and volume is called density.
is a measure of the amount of matter present in a given
volume of a substance. Density is normally expressed in units of
grams per cubic centimeter (g/cm
3
). In other words, density is the
mass in grams divided by the volume in cubic centimeters.
Density =
}
V
m
olu
as
m
s
e
}
D =
}
m
V
}
How would you find the density of 200 g of clay with a volume of
100 cm
3
? You calculate that the clay has a density of 200 g divided by
100 cm
3
, or 2 g/cm
3
. If you divide the clay in half and find the density
of one piece of clay, it will be 100 g/50 cm
3
, or 2 g/cm
3
—the same as
the original piece. Notice that density is a property of a substance that
remains the same no matter how much of the substance you have.
Density
Sample Problem
Practice the Math
Calculating Density
A glass marble has a volume of 5 cm
3
and a mass of 13 g. What is the
density of glass?
What do you know? Volume = 5 cm
3
, mass = 13 g
What do you want to find out? Density
Write the formula: D =
}
m
V
}
Substitute into the formula: D =
}
5
13
cm
g
3
}
Calculate and simplify: D = 2.6 g/cm
3
Check that your units agree: Unit is g/cm
3
.
Unit of density is g/cm
3
. Units agree.
Answer: D = 2.6 g/cm
3
1. A lead sinker has a mass of 227 g and a volume of 20 cm
3
. What is the
density of lead?
2. A glass of milk has a volume of 100 cm
3
. If the milk has a mass of 103 g,
what is the density of milk?
reading tip
The density of solids is
usually measured in grams
per cubic centimeter
(g/cm
3
). The density of
liquids is usually measured
in grams per milliliter
(g/mL). Recall that
1 mL 5 1 cm
3
.
Page 3 of 8

AB
44 Unit: Matter and Energy
Physical Changes
You have read that a physical property is any property that can be
observed without changing the identity of the substance. What then
would be a physical change? A is a change in any
physical property of a substance, not in the substance itself. Breaking
a piece of clay in half is a physical change because it changes only
the size and shape of the clay. Stretching a rubber band is a physical
change because the size of the rubber band changes. The color of the
rubber band sometimes can change as well when it is stretched.
However, the material that the rubber band is made of does not
change. The rubber band is still rubber.
What happens when water changes from a liquid into water vapor
or ice? Is this a physical change? Remember to ask yourself what has
changed about the material. Ice is a solid and water is a liquid, but both
are the same substance—both are composed of H
2
O molecules. As you
will read in more detail in the next section, a change in a substance’s
state of matter is a physical change.
Check Your Reading How is a physical change related to a substance’s
physical properties?
A substance can go through many different physical changes and
still remain the same substance. Consider, for example, the changes
that happen to the wool that ultimately becomes a sweater.
Wool is sheared from the sheep. The wool is then cleaned and
placed into a machine that separates the wool fibers from one
another. Shearing and separating the fibers are physical changes
that change the shape, volume, and texture of the wool.
The wool fibers are spun into yarn. Again, the shape and volume
of the wool change. The fibers are twisted so that they are packed
more closely together and are intertwined with one another.
The yarn is dyed. The dye changes the color of the wool, but it
does not change the wool into another substance. This type of
color change is a physical change.
Knitting the yarn into a sweater also does not change the wool into
another substance. A wool sweater is still wool, even though it no
longer resembles the wool on a sheep.
It can be difficult to determine if a specific change is a physical
change or not. Some changes, such as a change in color, also can occur
when new substances are formed during the change. When deciding
whether a change is a physical change or not, ask yourself whether you
have the same substance you started with. If the substance is the same,
then the changes it underwent were all physical changes.
4
3
2
1
physical change
MAIN IDEA WEB
As you read, organize your
notes in a web.
Page 4 of 8

The process of turning wool into a sweater requires
that the wool undergo physical changes. Changes in
shape, volume, texture, and color occur as raw wool
is turned into a colorful sweater.
Physical Changes
Preparing the wool produces physical
changes. The wool is removed from
the sheep and then cleaned before
the wool fibers are separated.
Further physical changes occur as
machine twists the wool fibers into
a long, thin rope of yarn.
Dyeing produces color changes
but does not change the basic
substance of the wool.
How does the yarn in the sweater differ
from the wool on the sheep?
Chapter 2: Properties of Matter 45
Shearing
1
Spinning
2
Dyeing
3
The final product, a wool
sweater, is still wool.
4
AB
Page 5 of 8

AB
46 Unit: Matter and Energy
INFER The bust of Abraham
Lincoln is made of bronze.
Why is the nose a different
color from the rest of the
head?
Chemical properties describe how
substances form new substances.
If you wanted to keep a campfire burning, would you add a piece
of wood or a piece of iron? You would add wood, of course, because
you know that wood burns but iron does not. Is the ability to burn a
physical property of the wood? The ability to burn seems to be quite
different from physical properties such as color, density, and shape.
More important, after the wood burns, all that is left is a pile of ashes
and some new substances in the air. The wood has obviously changed
into something else. The ability to burn, therefore, must describe another
kind of property that substances have—not a physical property but a
chemical property.
Chemical Properties and Changes
describe how substances can form new sub-
stances. Combustibility, for example, describes how well an object
can burn. Wood burns well and turns into ashes and other substances.
Can you think of a chemical property for the metal iron? Especially
when left outdoors in wet weather, iron rusts. The ability to rust is a
chemical property of iron. The metal silver does not rust, but eventually
a darker substance called tarnish forms on its surface. You may have
noticed a layer of tarnish on some silver spoons or jewelry.
The chemical properties of copper cause it to become a
blue-green color when it is exposed to air. A famous example
of tarnished copper is the Statue of Liberty. The chemical
properties of bronze are different. Some bronze objects tarnish
to a dark brown color, like the bust of Abraham Lincoln in the
photograph on the left.
Chemical properties can be identified by the changes they
produce. The change of one substance into another substance
is called a A piece of wood burning, an iron
fence rusting, and a silver spoon tarnishing are all examples of
chemical changes. A chemical change affects the substances
involved in the change. During a chemical change, combina-
tions of atoms in the original substances are rearranged to
make new substances. For example, when rust forms on iron,
the iron atoms combine with oxygen atoms in the air to form
a new substance that is made of both iron and oxygen.
A chemical change is also involved when an antacid
tablet is dropped into a glass of water. As the tablet dissolves,
bubbles of gas appear. The water and the substances in the
tablet react to form new substances. One of these substances
is carbon dioxide gas, which forms the bubbles that you see.
chemical change.
Chemical properties
RESOURCE CENTER
CLASSZONE.COM
Learn about the chemi-
cal properties of matter.
Page 6 of 8

AB
Not all chemical changes are as destructive as burning, rusting, or
tarnishing. Chemical changes are also involved in cooking. When you
boil an egg, for example, the substances in the raw egg change into
new substances as energy is added to the egg. When you eat the egg,
further chemical changes take place as your body digests the egg. The
process forms new molecules that your body then can use to function.
Check Your Reading Give three examples of chemical changes.
The only true indication of a chemical change is that a new
substance has been formed. Sometimes, however, it is difficult to tell
whether new substances have been formed or not. In many cases you
have to judge which type of change has occurred only on the basis
of your observations of the change and your previous experience.
However, some common signs can suggest that a chemical change has
occurred. You can use these signs to guide you as you try to classify a
change that you are observing.
What are some signs of a chemical change?
PROCEDURE
Measure 80 mL of water and pour it into one of the cups.
Add 3 full droppers of iodine solution. Record your observations.
Add 1 spoonful of cornstarch to the iodine solution and stir. Record
your observations.
Measure 50 mL of water and pour it into the second cup.
Using a clean eyedropper, add 4 full droppers of the iodine/cornstarch
solution to the second cup.
Drop a vitamin C tablet into the second cup and stir the liquid with a clean
spoon until the tablet is dissolved. Record your observations.
WHAT DO YOU THINK?
• What changes did you observe in the first cup?
in the second cup?
• Do you think that chemical changes occurred?
Why or why not?
• What are some characteristics of
chemical changes?
CHALLENGE Describe some chemical changes
that you have seen take place in your home or school.
6
5
4
3
2
1
Chemical Changes
Chemical Changes
SKILL FOCUS
Measuring
MATERIALS
• graduated
cylinder
• water
• 2 clear plastic
cups
• 2 eyedroppers
• iodine solution
• cornstarch
• spoon
• vitamin C tablet
TIME
15 minutes
47
Page 7 of 8

AB
48 Unit: Matter and Energy
Signs of a Chemical Change
You may not be able to see that any new substances have formed dur-
ing a change. Below are some signs that a chemical change may have
occurred. If you observe two or more of these signs during a change,
you most likely are observing a chemical change.
Production of an Odor Some chemical changes produce
new smells. The chemical change that occurs when an egg
is rotting produces the smell of sulfur. If you go outdoors
after a thunderstorm, you may detect an unusual odor in
the air. The odor is an indication that lightning has caused
a chemical change in the air.
Change in Temperature Chemical changes often are accom-
panied by a change in temperature. You may have noticed
that the temperature is higher near logs burning in a campfire.
Change in Color A change in color is often an indication
of a chemical change. For example, fruit may change color
when it ripens.
Formation of Bubbles When an antacid tablet makes
contact with water, it begins to bubble. The formation of
gas bubbles is another indicator that a chemical change may
have occurred.
Formation of a Solid When two liquids are combined, a solid called a
precipitate can form. The shells of animals such as clams and mussels
are precipitates. They are the result of a chemical change involving
substances in seawater combining with substances from the creatures.
Check Your Reading Give three signs of chemical changes. Describe one that you
have seen recently.
KEY CONCEPTS
1. What effect does observing a
substance’s physical properties
have on the substance?
2. Describe how a physical prop-
erty such as mass or texture
can change without causing
a change in the substance.
3. Explain why burning is a
chemical change in wood.
CRITICAL THINKING
4. Synthesize Why does the
density of a substance remain
the same for different amounts
of the substance?
5. Calculate What is the density
of a block of wood with a
mass of 120 g and a volume
of 200 cm
3
?
CHALLENGE
6. Infer Iron can rust when it
is exposed to oxygen. What
method could be used to
prevent iron from rusting?
Carbon dioxide bubbles
form as substances in the
tablet react with water.
Page 8 of 8
